{{ getSidebarNestedSelection() || 'Main menu' }}
- Respiratory
- HAI & Other Infectious Diseases
- Blood Virology, Women's Health, & Sexual Health
- TB & Emerging Infectious Diseases
- Oncology & Human Genetics
- Cepheid GeneXpert Systems
- GeneXpert® System Software
- Cepheid Connectivity and Interface Capabilities
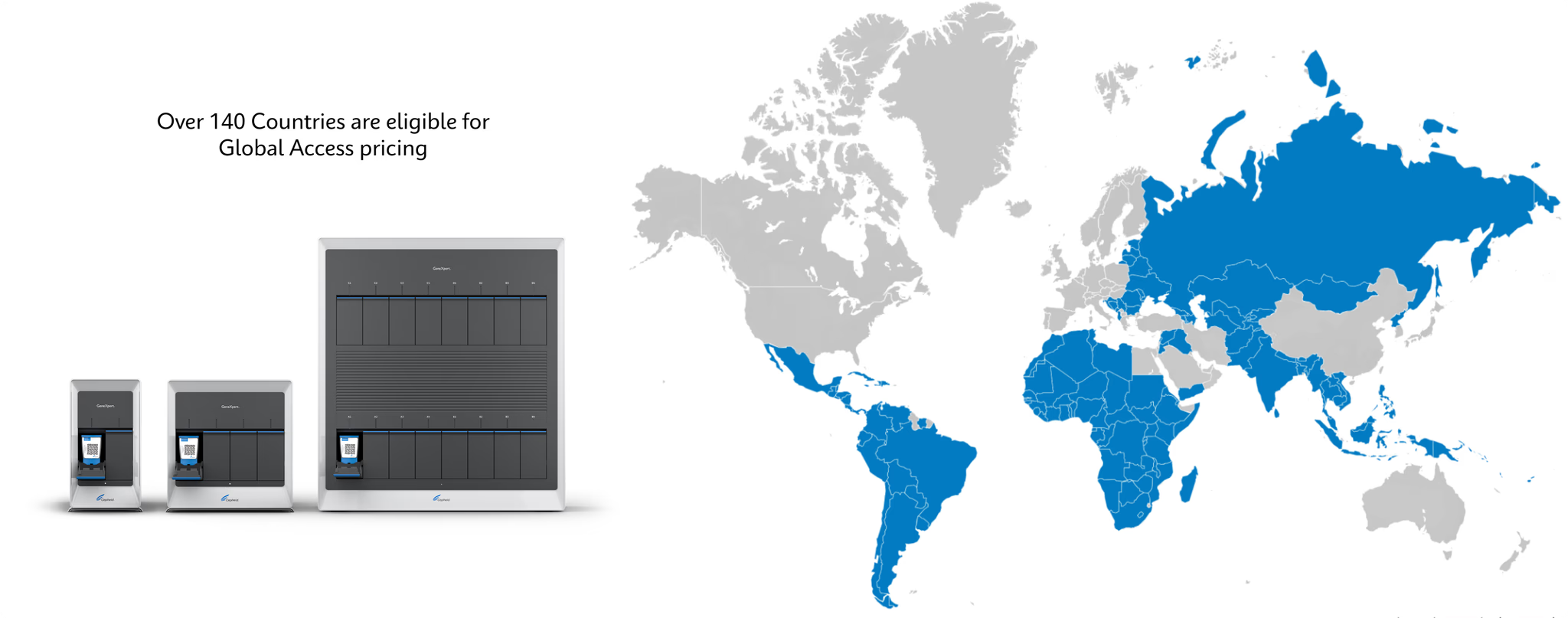
- Global Access
- Testing Everywhere
- Antibiotic Stewardship
- Customized Testing for Your Institution
- Knowing MORE Matters
- Xpert® SA Nasal Complete
- Xpert® MRSA/SA SSTI
- Xpert® C. difficile BT
- Xpert® Norovirus
- Xpert® MRSA/SA Blood Culture
- Xpert® vanA/vanB
- Xpert® MRSA NxG
- Xpert® Carba-R
- Xpert® HBV Viral Load
- Resistance Plus ® MG FleXible
- Xpert® Xpress GBS
- Xpert® CT/NG
- Xpert ® HIV-1 Qual XC
- Xpert® HCV VL Fingerstick
- Xpert ® HIV-1 Viral Load XC
- Xpert® TV
- Xpert® HPV v2
- Xpert® HCV Viral Load
- Xpert® Bladder Cancer Monitor
- Xpert® BCR-ABL Ultra p190
- Xpert® Bladder Cancer Detection
- Xpert® BCR-ABL Ultra
- Xpert® NPM1 Mutation