
Xpert® Xpress CoV-2 plus(10-test kit)
XPRS-COV2-10
Unit price
Subtotal
Product is not available for purchase in your region.
Xpert® Xpress CoV-2 plus


Xpert® Xpress CoV-2 plus(10-test kit)
XPRS-COV2-10
Unit price
Subtotal
Product is not available for purchase in your region.
Collection devices
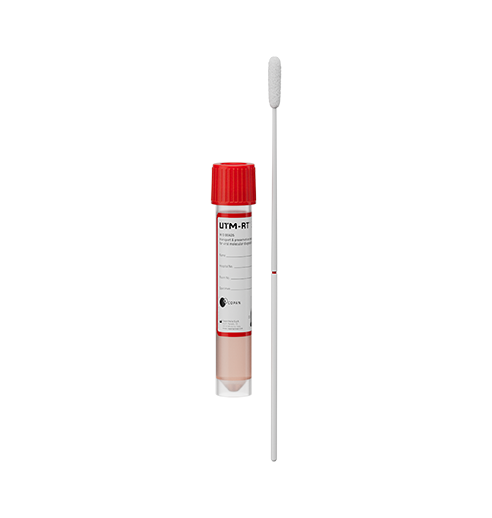
Copan 3C057N (Nasopharyngeal Swab) (Pack of 50)
3C057N
Unit price
Subtotal
Product is not available for purchase in your region.

Copan 3C064N (Nasal Swab) (Pack of 50)
3C064N
Unit price
Subtotal
Product is not available for purchase in your region.
Total
({{currency}})
0
Package Insert
eng
MSDS/SDS
eng
Verification Guide
eng
User Documentation
eng
Quick Reference Guide
eng
1. What sample types and collection materials can be used with Xpert Xpress CoV-2 plus?
1. What sample types and collection materials can be used with Xpert Xpress CoV-2 plus?
Sample Types: Nasopharyngeal and anterior nasal swab specimens collected from individuals with signs and symptoms of respiratory tract infection.
The specimen is collected and placed into a viral transport tube containing 3 mL viral transport medium (VTM)/Universal Transport Medium (UTM) or 2 mL eNAT™.
Refer to package insert for details.
2. What external quality control (QC) material options are available for Xpert Xpress CoV-2 plus?
2. What external quality control (QC) material options are available for Xpert Xpress CoV-2 plus?
The listed external controls are Zeptometrix External Controls:
These are described in the “materials available but not provided” section of the package insert.
3. Is the FDA cleared version of Xpert Xpress CoV-2 plus product also CLIA Waived?
3. Is the FDA cleared version of Xpert Xpress CoV-2 plus product also CLIA Waived?
Yes. The FDA cleared product is also CLIA Waived for use on GeneXpert® Xpress systems. Refer to applicable package insert for details.
4. Do you have any guidelines for verification studies of the Xpert Xpress CoV-2 plus test?
4. Do you have any guidelines for verification studies of the Xpert Xpress CoV-2 plus test?
Before going live with a new test, a verification study may be necessary. Please refer to your relevant laboratory accreditation regulations for verification requirements. A verification guide that describes the preparation of contrived positive specimens (prepared by spiking commercial reference material into negative specimens) is available for download on the Cepheid website: Verification Guide. The decision to perform verification studies and the level of verification required is at the discretion of the laboratory director and should be conducted according to applicable regulations.
5. What are my options for obtaining the ADF or the Package Insert for the Xpert® Xpress CoV-2 plus test?
5. What are my options for obtaining the ADF or the Package Insert for the Xpert® Xpress CoV-2 plus test?
In an effort to reduce waste, Cepheid will provide electronic copies of Instructions for Use (IFU) and Assay Definition Files (ADF). The IFUs, ADFs, and ADF import instructions for Xpert Xpress CoV-2 plus, are available for download here. A CD containing the ADF is also being provided with the first order of XPRS-COV2-10 or on their first order post ADF update for existing XPRS-COV2-10 users. For customers who would like to order additional copies, please fill out the CD ADF request form on the Cepheid website. USA CD ADF REQUEST FORM
6. Can I install the ADF of the EUA and FDA cleared versions concurrently on my GeneXpert?
6. Can I install the ADF of the EUA and FDA cleared versions concurrently on my GeneXpert?
Yes. For GeneXpert systems that are compatible with both the EUA and FDA-cleared products, the ADFs can be installed and used concurrently for both versions of the product, on the same computer. Please Note: The EUA version is not compatible with GeneXpert System with Touchscreen.
7. What are the intended use differences between the EUA and the FDA cleared version of the tests?
7. What are the intended use differences between the EUA and the FDA cleared version of the tests?
While there are no significant performance differences between both two versions of the tests, there are some differences in the product claims compared to the EUA version:
See IFU for details.
8. If I am already using the using the FDA cleared test (XPRS-COV2-10) in a Moderate Complexity environment: Is the ADF update mandatory, or can I continue to use my ADF?
8. If I am already using the using the FDA cleared test (XPRS-COV2-10) in a Moderate Complexity environment: Is the ADF update mandatory, or can I continue to use my ADF?
There are no design differences between the current Moderate Complexity version of ADF and new version of ADF with CLIA Waived extension. The new ADF allows the FDA cleared test to be run on our CLIA Waived systems. Therefore, the ADF update can be performed when customers are ready.
View our additional portals to find documents and resources not listed here.
View and download documents associated with your tests and instruments
View our self-help articles, create a case, or view contact information.
