

Frequently Asked Questions
Cepheid developed the Xpert® Xpress CoV-2 plus test with several enhancements compared to the Xpert® Xpress SARS-CoV-2. Key enhancements include:
- The addition of a 3rd gene target for SARS-CoV-2 (RdRP now included in addition to E and N2), to be more robust against mutations
- Improved Time to Result (TTR) at about 30 minutes
- Expansion of on-label transport media options to include the addition of eNAT®
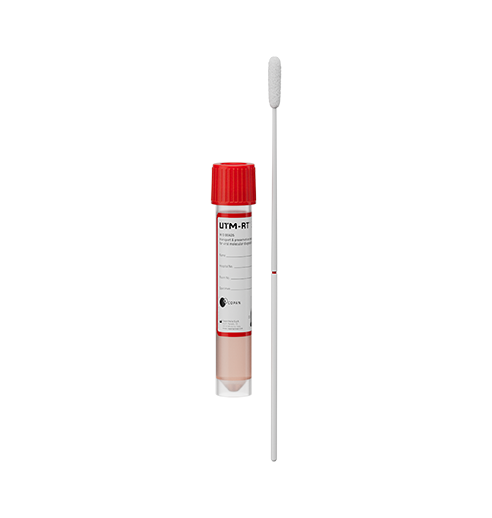
Sample Types:
GeneXpert Dx and Infinity Systems:
- From individuals suspected of COVID-19: Nasopharyngeal swab, anterior nasal swab, mid-turbinate swab, oropharyngeal swab or nasal wash/aspirate specimens
- From any individual, including individuals without symptoms or other reasons to suspect COVID-19: anterior nasal swab specimens
- In 3mL of viral transport media (UTM/VTM), 3mL of Saline, or 2mL of eNAT
GeneXpert Xpress systems (Hub and Tablet):
- From individuals suspected of COVID-19: Nasopharyngeal swab, anterior nasal swab, or mid-turbinate swabs specimens
- From any individual, including individuals without symptoms or other reasons to suspect COVID-19: anterior nasal swab specimens
- In 3mL of viral transport media (UTM/VTM), 3mL of Saline, or 2mL of eNAT.
Viral transport media are available from several commercial vendors who may use the terminology VTM (Viral Transport Medium) or UTM (Universal Transport Medium). Any brand meeting the guidelines for VTM/UTM can be used. UTM, VTM, Saline, and eNAT are on label.
For additional questions regarding SARS-CoV-2 Transport Media options, please refer to the FDA webpage FAQS on Viral Transport Media During COVID-19.
The listed external controls are Zeptometrix External Controls:
- SARS-Related Coronavirus 2 (SARS-CoV-2) External Run Control, Catalog# NATSARS(COV2)-ERC
- SARS Associated Coronavirus 2 (SARS-CoV-2) Negative Control, Catalog# NATSARS(COV2)-NEG
These are described in the “materials available but not provided” section of the package insert as the recommended source for external controls. Please consult with Cepheid’s Technical Support team to inquire about other commercial sources for QC material.
Before going live with a new test, a verification study may be necessary. Please follow your laboratory and regulatory requirements when instituting a new test in your laboratory. A verification guide that describes the preparation of contrived positive specimens (prepared by spiking commercial reference material into negative specimens) is available for download on this page.
In an effort to reduce our environmental footprint, Cepheid is moving towards CD-less products. The Package Inserts, ADFs and ADF import instructions for Xpert® Xpress CoV-2 plus are available for download on this page. For customers who prefer to obtain the ADFs in a CD, please fill out the CD ADF request using the following link:



