
XPRS4PLEX-10
On-demand 4-in-1 PCR test delivering actionable results with less than 1 minute of hands-on time. FDA cleared: moderate complexity and CLIA waived

XPRS4PLEX-10
On-demand 4-in-1 PCR test delivering actionable results with less than 1 minute of hands-on time. FDA cleared: moderate complexity and CLIA waived

XPRS-COV2-10
Designed with 3 gene targets for a rapid, robust detection of SARS-CoV-2

XPRSTREPA-10
Delivers accurate and timely results that optimizes fast treatment decisions

XPRSFLU-10
Qualitative detection and differentiation of influenza A and influenza B

XPRSFLU/RSV-10
Co-labeled Moderate Complexity and CLIA-Waived. Fast, accurate and reliable detection of Flu A, Flu B and/or RSV in one test

GXGI-10
Detection and identification of DNA and RNA from multiple bacteria, parasites and/or virus in ~ 74 minutes

GXSACOMP-10
Detection of S. aureus and MRSA in about 65 minutes

GXMRSA/SA-SSTI-10
Detect MRSA & SA Skin and Soft Tissue Infections in 62 minutes

GXNOV-10
Fast detection of Norovirus GI & GII with results available in 90 minutes

GXCDIFF/EPI-10
Detection and differentiation of C. difficile & the epidemic 027 strain in <45 minutes

GXMRSA/SA-BC-10
MRSA and S. aureus detection from gram-positive blood culture samples in 62 minutes

GXMRSA-NXG-10
Active MRSA Surveillance testing in around 70 minutes

GXVANA-10
Rapid VRE screening for active outbreak prevention and control in around 48 minutes

GXCARBAR-10
Detection and differentiation of KPC, NDM, VIM, IMP, and OXA-48 in 50 minutes

XPRSMVP-10
Multiplex vaginal panel that enables results within 60 minutes from a single specimen for Bacterial Vaginosis, Vulvovaginal Candidiasis, and Trichomoniasis

GXCT/NG-10
90-minute detection and differentiation of Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (NG)

GXGBSLBXC-10
Detection of Group B Streptococcus with dual target with results within one hour

GXTV-10
Fast molecular detection of Trichomonas vaginalis for both women and men.

XPRSGBS-10
Qualitative real-time PCR test providing accurate intrapartum detection of Group B Streptococcus in labor/delivery

GXHCV-SPAK
Point-of-care hepatitis C RNA test that enables same-day linkage to care

GXMPX-10
On-demand Mpox testing provides qualitative point-of-care results in approximately 36 minutes, with EAT results as soon as 25 minutes. For use under an Emergency Use Authorization in the United States

GXMTB/RIF-US-10
Accurate Detection of MTB and Rifampin Resistance Mutations

GXFIIFV-10
30-Minute Test for Genetic Risk of Thrombosis

GXBCRABL-US-10
Delivering MORE confidence with fast, accurate Chronic Myeloid Leukemia molecular monitoring results



Available in 4, 16, 48, or 80-module configurations, each system has Cepheid’s proven GeneXpert® module at its analytic core and uses our patented cartridge technology for every Xpert® test. The platform is standardized yet scalable across the healthcare continuum — from hospital labs to emergency departments and decentralised settings.

Article
4m Read
May 19, 2025
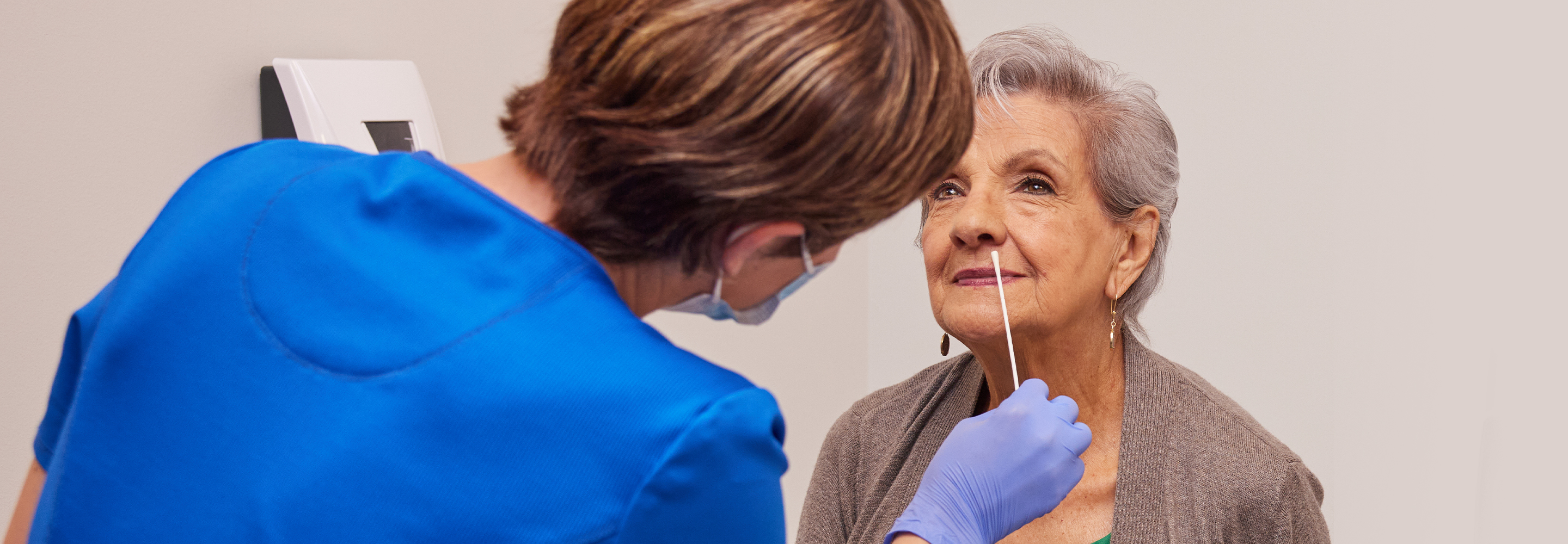
RESPIRATORY HEALTH

Article
5m Read
January 07, 2026
TECH AND DISEASE TRENDS
View our additional portals to find documents and resources not listed here.
View and download documents associated with your tests and instruments
View our self-help articles, create a case, or view contact information.
