
XPRSTREPA-CE-10
Delivers accurate and timely results that optimize fast treatment decisions

XPRSTREPA-CE-10
Delivers accurate and timely results that optimize fast treatment decisions

XPRSFLU/RSV-CE-10
Rilevamento accurato e affidabile di influenza A, influenza B e RSV per la rapida somministrazione di terapie mirate.


GXSACOMP-CE-10
Detection of S. aureus and MRSA in about 65 minutes

GXMRSA/SA-SSTI-CE
Detect MRSA & SA Skin and Soft Tissue Infections in 62 minutes

GXCDIFFBT-CE-10
Detection of Clostridioides difficile infection with an independent call-out of binary toxin and differentiation of the 027 strain in around 45 minutes

GXNOV-CE-10
Fast detection of Norovirus GI & GII with results available in 90 minutes


GXMRSA/SABC-CE-10
MRSA and S. aureus detection from gram-positive blood culture samples in 62 minutes

GXVANA/B-CE-10
Rapid VRE screening for active prevention, control, and outbreak management in around 48 minutes

GXMRSA-NXG-CE-10
Sorveglianza attiva di MRSA in circa 70 minuti.


GXCARBARP-CE-10
Rilevamento e differenziazione di KPC, NDM, VIM, IMP-1 e OXA-48 in 50 minuti

GXHBV-VL-CE-10
Quantitation of Hepatitis B Virus (HBV) in less than one hour

S2A-95004
Identificazione di M. genitalium e dell’eventuale resistenza ai macrolidi

XPRSGBS-CE-10
Qualitative real-time PCR test providing accurate intrapartum detection of Group B Streptococcus in Labor/delivery

GXCT/NGX-CE-10
90-minute detection and differentiation of Chlamydia trachomatis (CT) and Neisseria gonorrhoeae

GXHIV-QA-XC-CE-10
A molecular point-of-care test that delivers results in around 90 minutes

GXGBSLBXC-10
Detection of Group B Streptococcus with dual target with results within one hour

GXHCV-FS-CE-10
Detection and quantification of Hepatitis C virus (HCV) in about an hour

GXHIV-VL-XC-CE-10
Test molecolari di nuova generazione per il monitoraggio della carica virale e dell’infezione da HIV-1.

GXTV-CE-10
Fast molecular detection of Trichomonas vaginalis for both women and men.

GXHPV2-CE-10
Rilevamento affidabile del DNA di HPV ad alto rischio con genotipizzazione di HPV 16 e 18/45

GXHCV-VL-CE-10
Highly sensitive, timely results to support patient management


GXHCV-SPAK
Point-of-care hepatitis C RNA test that enables same-day linkage to care

GXMTB/XDR-10
Semplificazione dei test di farmaco sensibilità per la TB utilizzando una maggiore capacità di multiplex con la tecnologia GeneXpert® a 10 colori

GXMTB/RIF-ULTRA-50
Detection of Mycobacterium tuberculosis complex and Rifampin-resistance associated mutations in less than 80 minutes



GXBLAD-CM-CE-10
Monitoraggio qualitativo delle recidive in pazienti con precedente diagnosi di tumore della vescica in circa 90 minuti.

GXFIIFV-10
Test del rischio genetico di trombosi in 30 minuti.

GXFFPE-LYSIS-CE-10
Valutazione standardizzata basata su mRNA dei biomarcatori ESR1, PGR, ERBB2 e MKi67 del cancro della mammella in meno di due ore.

GXBLAD-CD-CE-10
Rilevamento della presenza del tumore della vescica in pazienti con ematuria in circa 90 minuti.

GXBCRABL-10
Monitoraggio sensibile e quantitativo dell'mRNA di BCR-ABL in pazienti affetti da leucemia mieloide cronica (LMC).



Available in 4, 16, 48, or 80-module configurations, each system has Cepheid’s proven GeneXpert® module at its analytic core and uses our patented cartridge technology for every Xpert® test. The platform is standardized yet scalable across the healthcare continuum — from hospital labs to emergency departments and decentralised settings.

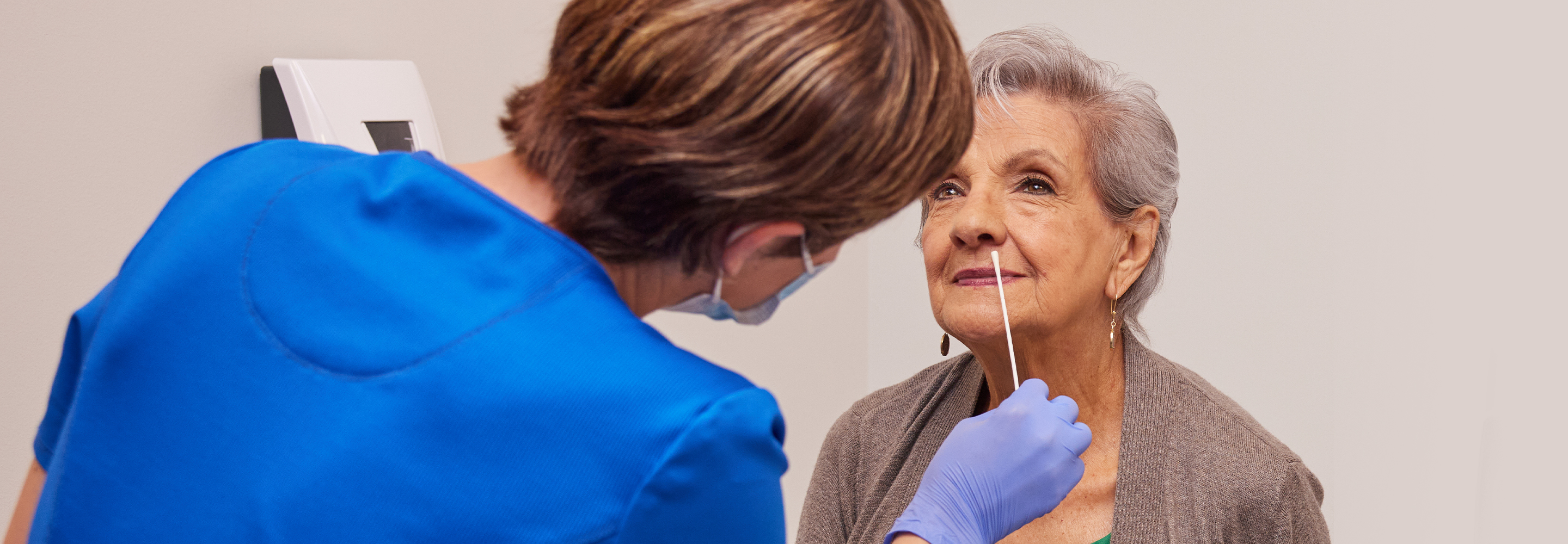
Articolo
4 minuti di lettura
19 maggio 2025
SALUTE RESPIRATORIA

Prospettiva dell’esperto
5 minuti di lettura
14 maggio 2025
COMUNITÀ E SALUTE GLOBALE
View our additional portals to find documents and resources not listed here.
View our self-help articles, create a case, or view contact information.
