
XPRSTREPA-CE-10
Delivers accurate and timely results that optimize fast treatment decisions

XPRSTREPA-CE-10
Delivers accurate and timely results that optimize fast treatment decisions

XPRSFLU/RSV-CE-10
Präziser und zuverlässiger Nachweis von Influenza A, Influenza B und RSV für zeitnahe, zielgerichtete Therapieentscheidungen.


GXSACOMP-CE-10
Detection of S. aureus and MRSA in about 65 minutes

GXMRSA/SA-SSTI-CE
Detect MRSA & SA Skin and Soft Tissue Infections in 62 minutes

GXCDIFFBT-CE-10
Detection of Clostridioides difficile infection with an independent call-out of binary toxin and differentiation of the 027 strain in around 45 minutes

GXNOV-CE-10
Fast detection of Norovirus GI & GII with results available in 90 minutes


GXMRSA/SABC-CE-10
MRSA and S. aureus detection from gram-positive blood culture samples in 62 minutes

GXVANA/B-CE-10
Rapid VRE screening for active prevention, control, and outbreak management in around 48 minutes

GXMRSA-NXG-CE-10
Aktive MRSA-Surveillance-Testung in etwa 70 Minuten


GXCARBARP-CE-10
PCR-Nachweis und Differenzierung der Carbapenem-Resistenzgene KPC, NDM, VIM, IMP-1 und OXA-48 in 50 Minuten.

GXHBV-VL-CE-10
Quantitation of Hepatitis B Virus (HBV) in less than one hour

S2A-95004
M. genitalium + Nachweis von Makrolid-Resistenz

XPRSGBS-CE-10
Qualitative real-time PCR test providing accurate intrapartum detection of Group B Streptococcus in Labor/delivery

GXCT/NGX-CE-10
90-minute detection and differentiation of Chlamydia trachomatis (CT) and Neisseria gonorrhoeae

GXHIV-QA-XC-CE-10
A molecular point-of-care test that delivers results in around 90 minutes

GXGBSLBXC-10
Detection of Group B Streptococcus with dual target with results within one hour

GXHCV-FS-CE-10
Detection and quantification of Hepatitis C virus (HCV) in about an hour

GXHIV-VL-XC-CE-10
Molekularer Test der nächsten Generation zur Überwachung von Viruslast und HIV-1-Infektionen

GXTV-CE-10
Fast molecular detection of Trichomonas vaginalis for both women and men.

GXHPV2-CE-10
Zuverlässiger Nachweis von Hochrisiko-HPV-DNA mit Genotypisierung von HPV 16 und 18/45

GXHCV-VL-CE-10
Highly sensitive, timely results to support patient management


GXHCV-SPAK
Point-of-care hepatitis C RNA test that enables same-day linkage to care

GXMTB/XDR-10
Vereinfachung der TB-Arzneimittelempfindlichkeitstests durch erhöhte Multiplexfähigkeit mit 10 Farben GeneXpert®-Technologie

GXMTB/RIF-ULTRA-50
Detection of Mycobacterium tuberculosis complex and Rifampin-resistance associated mutations in less than 80 minutes



GXBLAD-CM-CE-10
Qualitatives Monitoring auf Rezidive bei Patient/innen, bei denen bereits früher Blasenkrebs diagnostiziert wurde, in rund 90 Minuten

GXFIIFV-10
30-minütiger Test zur Bestimmung des genetischen Thromboserisikos

GXFFPE-LYSIS-CE-10
Standardisierte Bestimmung der Brustkrebs-Biomarker ESR1, PGR, ERBB2 und MKi67 auf mRNA-Ebene in weniger als zwei Stunden

GXBLAD-CD-CE-10
Nachweis des Vorliegens von Blasenkrebs bei Patient/innen mit Hämaturie in rund 90 Minuten

GXBCRABL-10
Standardisierte Messung und quantitatives Monitoring der BCR-ABL-mRNA bei Patienten mit chronischer myeloischer Leukämie (CML) in weniger als 2 Stunden



Available in 4, 16, 48, or 80-module configurations, each system has Cepheid’s proven GeneXpert® module at its analytic core and uses our patented cartridge technology for every Xpert® test. The platform is standardized yet scalable across the healthcare continuum — from hospital labs to emergency departments and decentralised settings.

Cepheids Zukunftsvision lautet: „Nahtloser Zugang zur Diagnostik in einer vernetzten Welt.“ Wir schließen Partnerschaften, um noch besser Ihre Herausforderungen zu verstehen. Zudem arbeiten wir kontinuierlich an der Weiterentwicklung der Konnektivitätslösungen, damit sie noch einfacher und intuitiver zu bedienen sind und Ihnen eine noch bessere Patientenversorgung ermöglichen.
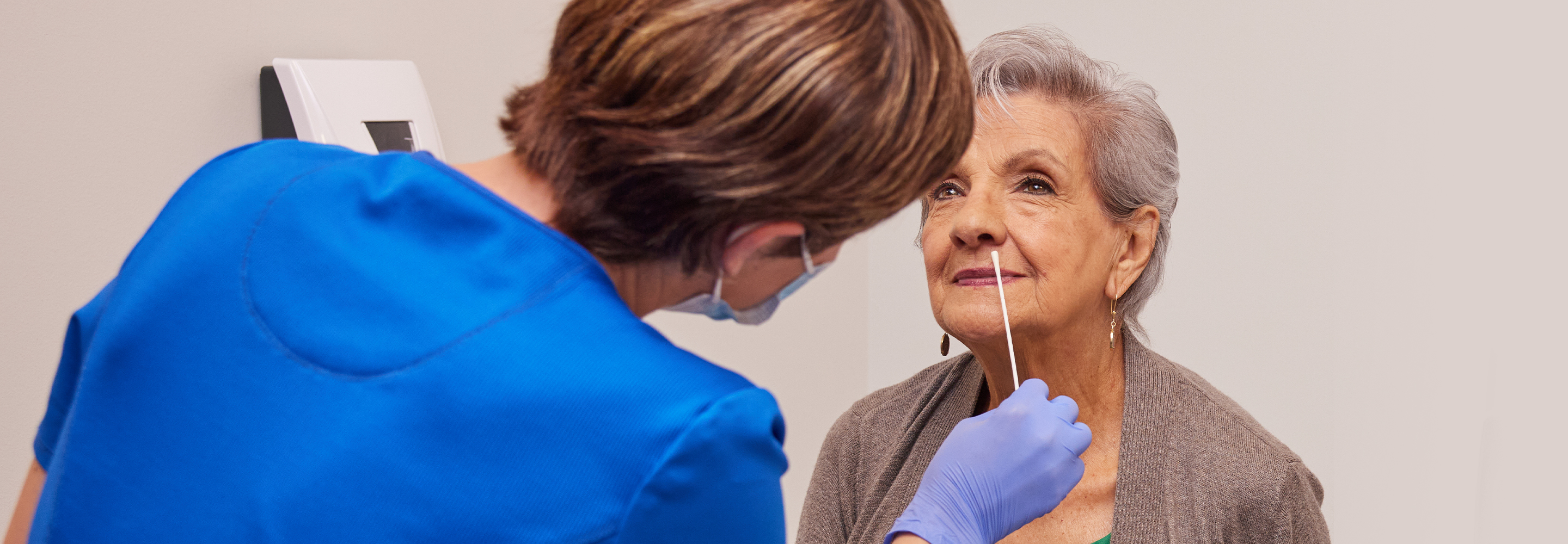
Artikel
Lesedauer von 4 Min.
19. Mai 2025
ATEMWEGSGESUNDHEIT

Experten-Perspektive
Lesedauer von 5 Min.
14. Mai 2025
GEMEINSCHAFT UND GLOBALE GESUNDHEIT
View our additional portals to find documents and resources not listed here.
View our self-help articles, create a case, or view contact information.
