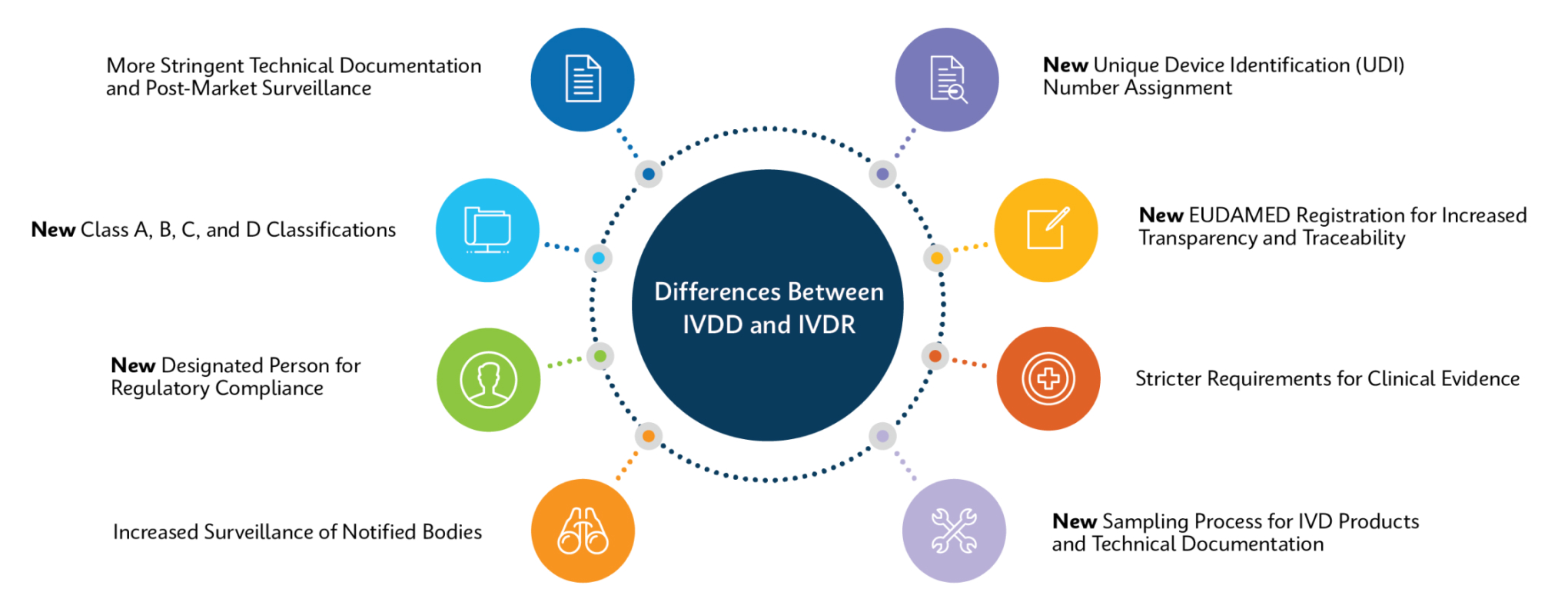
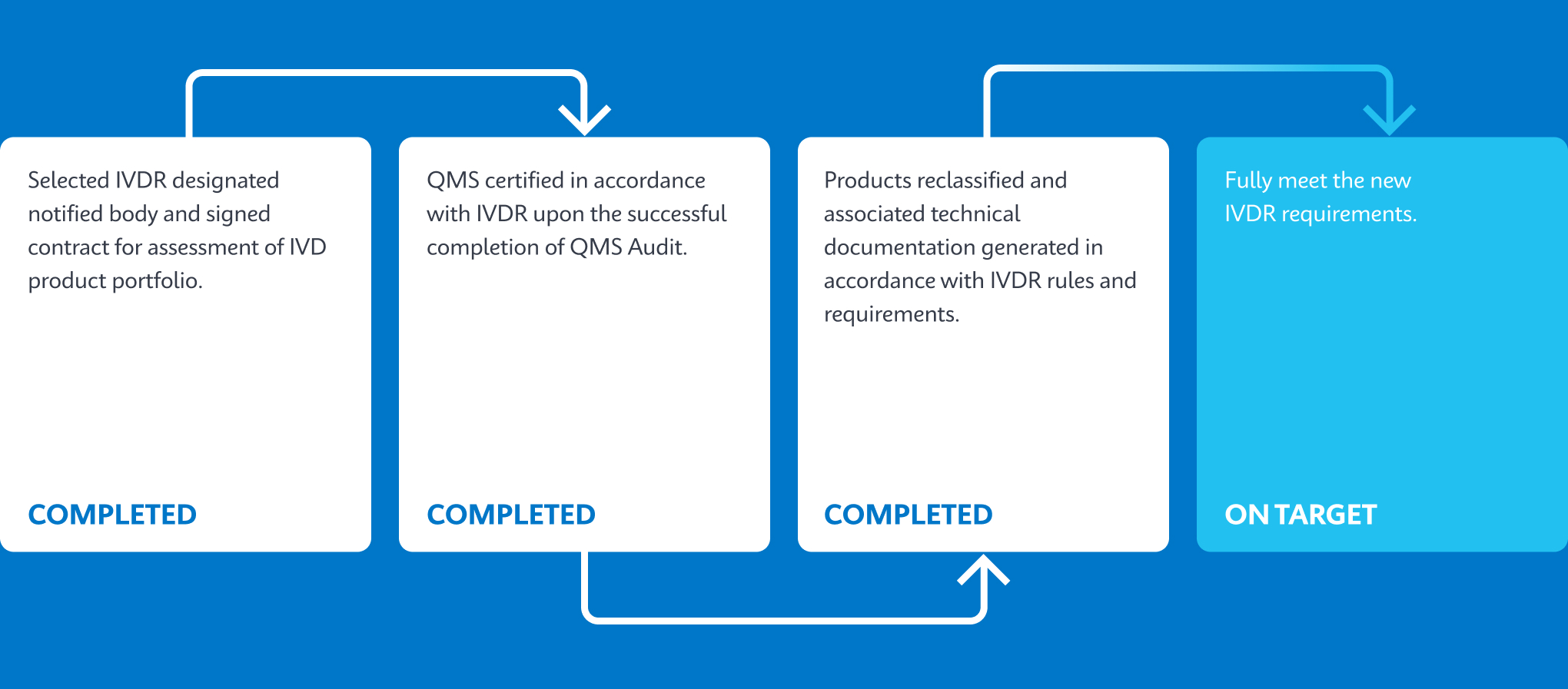
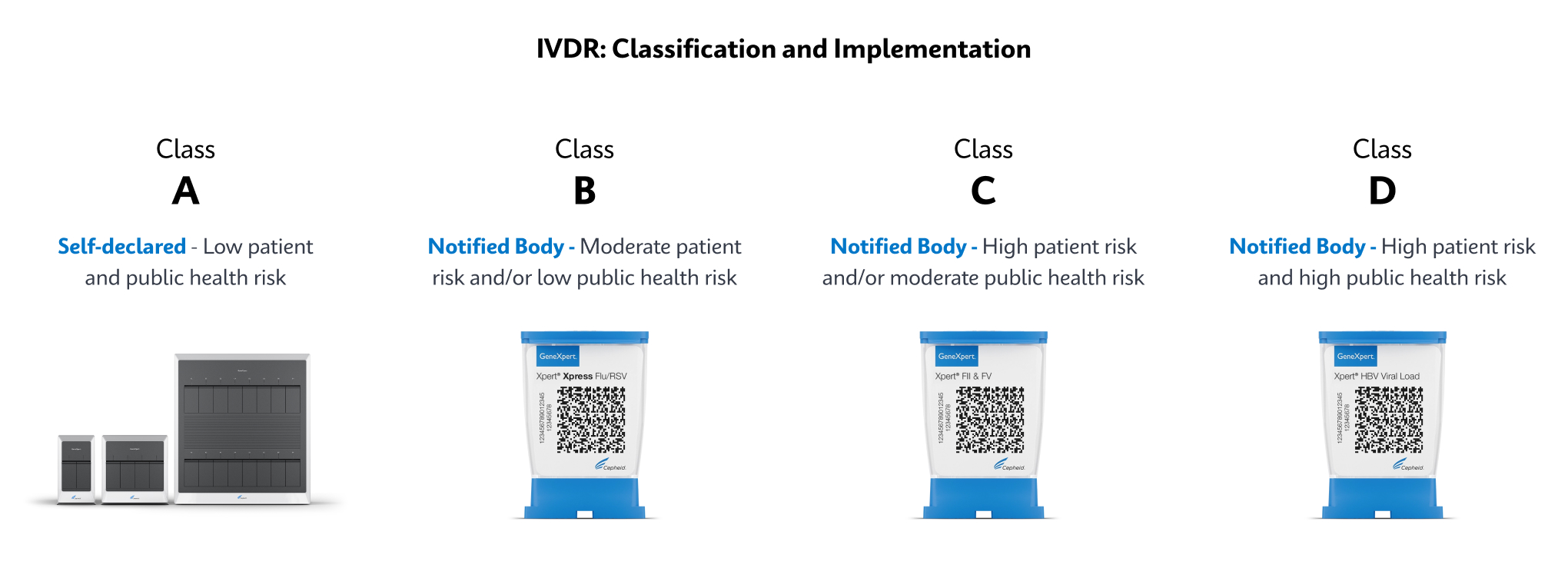
How Does IVDR Impact Customers?
Cepheid is working to ensure all relevant products are IVDR-compliant and have processes in place for recertification to ensure product continuity for all our customers.
For most products, changes will be minimal: same product code, same performance, and validation requirements will be kept to a minimum. For a few products, there will be a change in catalog number, which may entail more extensive update/validation processes.
Changes may include but not limited to:
- For devices that require notified body oversight under IVDR, IFU changes and Xpert test kits will include the CE Notified Body Number alongside the CE mark
- Intended purpose statements
- Warnings or contraindications
- Additions or alterations to sample types, stability, interfering substances, linearity, reference ranges, specificity, and sensitivity
- Updates to supporting references
- Changes to product labeling
Before a product is launched, all users will receive a letter detailing these changes and Cepheid's teams will be mobilized to help make this changeover as smooth as possible.